Introduction
Electrochemistry is a branch of chemistry concerned with the interconversion of chemical energy into electrical energy and vice-versa. To carry out these conversions, different types of electrochemical cells are used.
Electrochemical cells are of two types, which are described below:
- Galvanic cell/ Voltaic cell/ Daniell cell – In this type of electrochemical cell, a spontaneous chemical generates an electric current.
- Electrolytic cell – In this type of electrochemical cell, an electric current drives a non-spontaneous chemical reaction.
Daniell cell construction/working –
In one beaker, on the left-hand side, the Zn rod is dipped in a Zinc sulfate solution. In another beaker, on the right-hand side, the Cu rod is dipped in a copper sulfate solution. The two rods are connected using a conducting wire. In a spontaneous chemical reaction that occurred on the Zn rod, Zn was oxidized. Zn ions were mixed into the solution, and electrons (negative charge) accumulated at the rod. And those electrons accumulated and travelled to the Cu rod, causing the reduction of copper sulfate solution. This process continued, and the electrons flowed from Zn to Cu, whereas the current flowed in the opposite direction, i.e., from Cu to Zn.
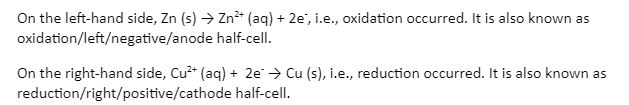
After some time, in the Daniell cell, a problem arose. In the left half cell, Zn2+ ions were getting accumulated in the solution, but then, the concentration of positive charge increased in the solution, and they started to pull the electrons on the rod towards itself, which restricted the outward flow of electrons from the Zn rod to the Cu rod. Similarly, in the right half-cell, the concentration of sulfate ions increased in the solution, which further restricted the inward flow of electrons.
This issue was resolved by using a salt bridge. It is an inverted U-shaped tube filled with an inert electrolyte, such as KCl, KNO3, K2SO4 in an agar-agar solution. Salt bridge provides a negative charge of inert electrolyte to the left half cell and neutralizes the excess positive charge, whereas it provides a positive charge of inert electrolyte to the right half cell and neutralises the excess negative charge. Hence, it maintains the neutrality of half-cells and completes the circuit by allowing the flow of electrons.

There are two conditions for the electrolyte to be used in the salt bridge:
- It should be inert. It should not react and form precipitates with any of the ions of the half-cells.
- The ions of the electrolyte should have good mobility.
Functions of salt bridge:
- It connects the solution of two half cells to complete the circuit.
- It minimizes the liquid junction potential which is potential difference between the junction of two liquids.
- It maintains electric neutrality of the solution in order to give continuous flow or generation of current.
- If the salt bridge is removed then voltage drops to zero.
- It prevents mechanical mixing of two electrolytic solutions.
Representation of Daniell Cell –
The anode is written on the left-hand side and the cathode on the right-hand side. The vertical line denotes the contact between the two phases. The cell’s anode is represented first by writing the metal and then by the metal ion present in the electrolytic solution. Both are separated by a vertical line. The molar concentration is written in brackets after the formula of ion.

Electrolytic Cells –
Another type of electrochemical cell is an electrolytic cell. These cells are the opposite of electrochemical cells. Here, the electric current is used to carry out chemical reactions.
In a container carrying electrolytic solution, anode and cathode are attached using a battery. The anode is connected to the positive half of the battery, whereas the cathode is connected to the negative half of the battery. Upon supplying current, the ions move towards electrodes of opposite polarity and undergo oxidation and reduction. Here, the anode is positively charged and, hence, reduction takes place. The cathode is negatively charged, and oxidation takes place. The redox reaction taking place at an electrolytic cell is non-spontaneous and takes place only when an electric current is supplied.
Conclusion
Electrochemical cells and their types are integral in the chemical industry and our day-to-day lives. Battery, which is used in remote controllers, watches, etc., is one example. Galvanic cells use energy generated by spontaneous chemical reactions to produce electric current, whereas in electrolytic cells, supplied electric current facilitates non-spontaneous reactions.
 Profile
Profile Settings
Settings Refer your friends
Refer your friends Sign out
Sign out





