The dual nature theory of light was proposed by Albert Einstein in 1905. The theory says that light acts as both a particle and a wave. The energy associated with each particle is called a photon. This quantum of energy of photons is expressed in terms of Planck’s equation. The hypothesis presented by Einstein helped to explain that light is emitted when a current passes through a gas in a cathode ray tube. An atom is said to be at a ground point where it has very low energy. As the current passes at a lower rate the atomic force increases. In a situation where the atom is stronger than the ground state, it is said to be excited. In this case, the atom is unstable and, eventually, will return to a state of normalcy and in this process, energy is lost in the form of electromagnetic radiation. When light is transmitted to a prism then it splits into different light bands in certain waves and these bands are called spectrum emitted by the line of that particular object.
Theories on dual nature of matter:
1. De Broglie’s hypothesis:
In 1924, Lewis de Broglie, physicist, said all objects whether microscopic or macroscopic possess dual nature i.e., both particle and wave. The wave associated with the particle is known as the Matter-wave or de Broglie’s wave. In other words, moving matter shows wave-like properties such as diffraction and interference and when it is resting it shows the properties of the particle.
The wavelength associated with the particle of a wave is calculated as:
The energy of the photon is given by
E = hv ——- 1
Where v = frequency of a wave
h = Planck’s constant
For particle character of photon, the energy is given by:
E = mc2 ——— 2
Where m = mass of photon
c = velocity of light
Equating eqn. 1 and 2
hv = mc2
here c = vλ and v = c/λ
Therefore, substituting for v
hc/λ = mc2
h = mcλ
λ = h/mc
The above equation is called de Broglie’s Equation and λ is known as de Broglie’s wavelength.
2. Heisenberg’s Uncertainty Principle:
The momentum and position of a particle are not possible to determine simultaneously. Mathematically, it is expressed as ∆ x ∆P ≥ (h / 4π).
Where ∆x is the Uncertainty in position
∆P is the Uncertainty in Momentum.
3. Planck’s Quantum Theory:
According to Planck’s Quantum theory, when heat is applied to a black body, it leads to the emission of thermal radiation with different wavelengths. Planck’s quantum theory postulates are:
- Energy is either absorbed or radiated by matter in discrete quantities, in the form of small packets or bundles.
- The smallest packet of energy is known as quantum. A quantum of light is known as a photon.
- The frequency of radiation is directly proportional to the energy of quantum emitted or absorbed.
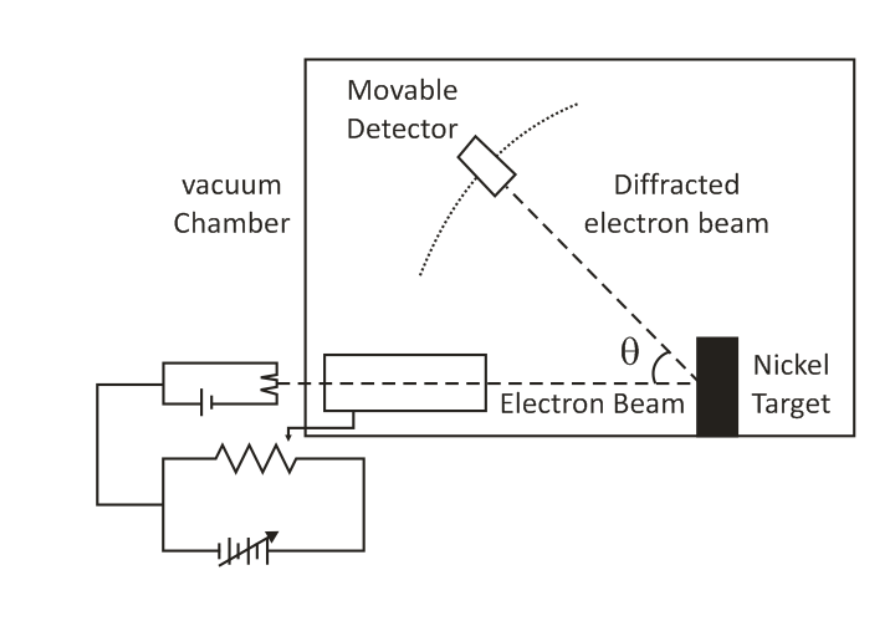
4. Davison and Germer’s Experiment:
When a beam of electrons falls on the surface of nickel crystal, the scattered or reflected rays are received on a photographic plate and a diffraction pattern is obtained as that of x-ray. Since x-ray is electromagnetic waves, they are confirmed to have wave character. Therefore, electrons must also have a wave character.
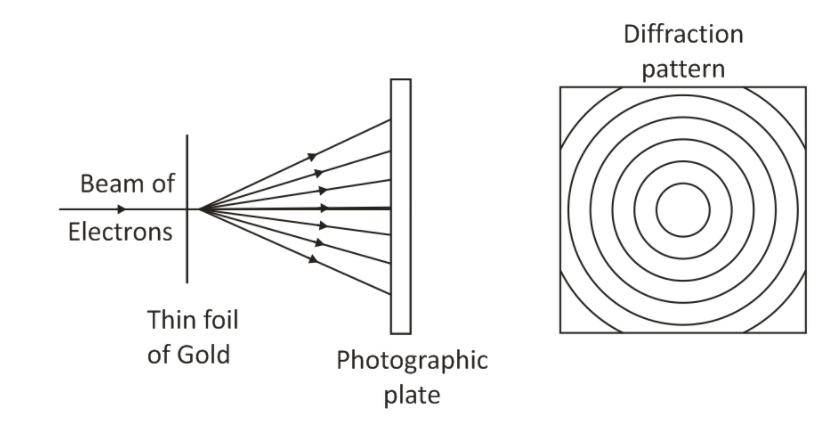
5. Thomson’s experiment:
The experiment was conducted with a thin foil of gold instead of nickel as in Davisson’s experiment. When a beam of electrons passes through a thin foil of gold and is received on a photographic plate, a diffraction pattern is observed.
Conclusion:
Through the work of Niels Bohr, Albert Einstein, Arthur Compton, Max Planck, Louis de Broglie and many other scientists, the current scientific theory holds the fact that all particles reflect the nature of waves and vice versa. Light properties and matter indicate that both light and matter have a dual nature of wave particles. While one thing to keep in mind is that wave structures and particles are fundamental.
 Profile
Profile Settings
Settings Refer your friends
Refer your friends Sign out
Sign out





