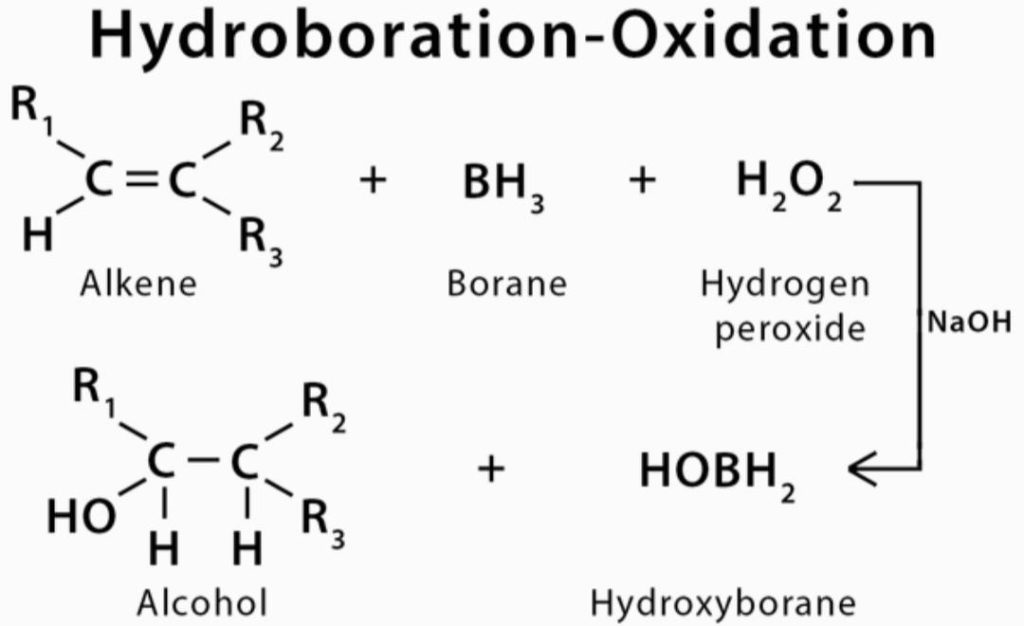
The hydroboration oxidation reaction is an organic chemical reaction that is used to convert alkenes into neutral alcohols. It is a type of oxidation reaction. This is accomplished through a two-step process that consists of a hydroboration step and an oxidation step, respectively. This is accomplished through a net addition of water (across the entire double bond).
The mechanism of the hydroboration oxidation reaction can be thought of as an anti-Markovnikov reaction, in which a hydroxyl group attaches itself to the carbon that is less substituted than the other carbons. According to Herbert Charles Brown, an English-born American chemist who was born in New York in 1927, the hydroboration oxidation reaction was discovered in the second half of the 1950s. He went on to receive the Nobel Prize in Chemistry in 1979 for his contributions to this field.
The example of hydroboration oxidation reaction is given below,
Hydroboration steps:
The addition of borane in the form of BH3 to the given double bond is the first step in the procedure. This results in the transfer of a hydrogen atom to the carbon atom that is adjacent to the carbon atom that is bonded to the boron atom as a result of the reaction. The hydroboration step described above is now repeated twice more, resulting in three alkenes that are attached to the boron atom from the initial BH3 that were formed.
Trialkyl borane is the name given to the compound formed by the reaction of three alkenes with borane to form a cyclic compound. Using a base (or water) and hydrogen peroxide, this trialkyl borane is now being treated further. Carbon-OH group bonds are formed as a result of the replacement of boron-carbon bonds. It is also possible to observe the transformation of boron into boric acid.
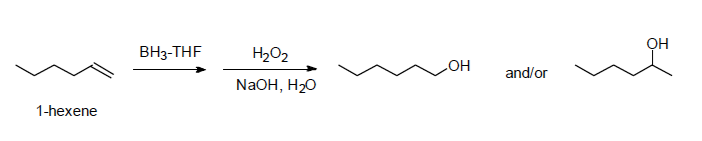
This method, which is illustrated below, can be used to convert 1-hexene into 1-hexanol.
Hydroboration-oxidation for Alkynes mechanism:
Additionally, hydroboration can occur with the terminal alkynes. This is also carried out in an anti-Markovnikov manner as well. The less substituted carbon atom, which is also the least hindered, becomes a priority target for the boron atom’s attack because it is less hindered. It is necessary to use a large amount of borane in order to stop the reaction at the alkenyl group attached to the borane stage. If borane is used alone, it will result in the hydroboration of both of the alkyne’s pi-bonds if the alkyne is used alone.
The oxidation step of this process begins with the oxidation of the alkyl borane to vinyl alcohol, which contains both an alkene group and an OH group.
Due to hydroxide reactions occurring in the basic solution, this oxidation occurs. This alcohol is now subjected to tautomerization, which results in the formation of a stable aldehyde form.
Is Hydroboration Oxidation Syn or anti:
The addition of a hydrogen and a hydroxyl group in the same location as the double bond is the result of this method of construction. The anti-Markovnikov response of hydroboration-oxidation is characterised by the addition of the less-substituted carbon to the hydroxyl ring.
In most cases, hydrogenation occurs after syn-addition, but there are some circumstances in which anti-addition is more likely.
Conclusion:
The hydroboration–oxidation reaction is a two-step hydration reaction in which an alkene is converted into an alcohol in one step. The process results in the simultaneous addition of a hydrogen and a hydroxyl group to the site where the double bond had previously existed.
The mechanism of the hydroboration oxidation reaction can be thought of as an anti-Markovnikov reaction, in which a hydroxyl group attaches itself to the carbon that is less substituted than the other carbons.
 Profile
Profile Settings
Settings Refer your friends
Refer your friends Sign out
Sign out




