Around the central chlorine atom, chlorine trifluoride contains five electron density zones (3 bonds and two lone pairs). They are organised in a trigonal bipyramidal form with a 175° F(axial)-Cl-F(axial) bond angle.ClF3 is colourless as gas and turns into a pale green-yellow liquid when it condenses. It is a highly reactive, toxic, and caustic substance. Since its discovery, a variety of applications has been using chlorine trifluoride.
Because they occupy more space than the bonds, the two lone pairs adopt equatorial positions. A T-shaped molecule emerges as a result of this process.
Chlorine Trifluoride (CIF3)
The chemical formula ClF3 represents chlorine Trifluoride. It is an interhalogen substance.
ClF3 was initially observed as a result of fluorinating chlorine. Chlorine and fluorine-based halides are formed when it reacts with metals. Phosphorus Trichloride (PCl3) and Phosphorus Pentafluoride are two examples (PF5). It produces Hydrogen Chloride or Hydrogen Fluoride when it reacts aggressively with water. There is also a release of oxygen.
Chlorine trifluoride is primarily utilised as a cleaning agent in the semiconductor sector. It has been borne in mind for use as a storable oxidizer in rockets. Concerns about storage, on the other hand, have not been addressed.
ClF3 is a solid oxidising and fluorinating agent that is also hypergolic. It is frequently flammable and, when mishandled, can cause serious harm. When dealing with hazardous substances, it is critical to take all necessary measures.
Properties of chlorine trifluoride
- The chemical formula of chlorine trifluoride is CIF3
- CIF3 has a T-shaped molecular geometry.
- Trigonal bipyramidal is the electron geometry of CIF3
- Hybridisation of CIF3 is sp3d
- There are two lone pairs in CIF3
- The formal charge of ClF3 is 0.
- It corrodes metals and tissues.
- At 53°F, it boils.
- It has a high oxidising and fluorinating agent
- ClF3 has a melting point of 76.34 degrees Celsius and a boiling point of 11.75 degrees Celsius.
- Its molecular mass is 92.45 gmol-1.
Valence electrons of chlorine trifluoride
Chlorine Trifluoride is made up of three Fluorine atoms and one chlorine atom.
Chlorine, which belongs to group 7 of the periodic table, has seven valence electrons and a valency of -1. The electrical configuration of chlorine is [Ne]3s23p5.
As a result, the valence electrons contributed by the chlorine atom are 7 x 1 = 7.
Fluorine belongs to Periodic Group 17 and has the electrical configuration [He] 2s22p5. As a result, the three Fluorine atoms contribute 7 x 3 = 21 Valence Electrons.
As a result, the total number of valence electrons in ClF3 is calculated as follows:
Valence Electrons: 7[Cl] + 21[F] = 28
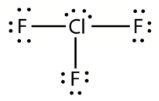
Lewis structure
ClF3 has the following Lewis Structure:
CIF3 Hybridisation
The centre Chlorine atom pulls three Fluorine atoms together to form Chlorine Trifluoride. The Lewis structure of the ClF3 molecule determines the hybridisation.
On the central Chlorine atom, there are two lone pairs. Chlorine also forms covalent connections with the Fluorine atoms in its environment.
There are three Cl-F bonds and two lone electron pairs in total.
The core Chlorine atom requires three unpaired electrons to form bonds with Fluorine.
The hybridisation process involves one 3s orbital, three 3p orbitals, and one 3d orbital. Five sp3d orbitals are formed as a result of this.
As a result, the central Chlorine atom in Chlorine Trifluoride is sp3d hybridised.
ClF3 bond angles
According to the VSEPR hypothesis, the constituent atoms repel each other due to the presence of lone pairs. ClF3 has 87.5-degree bond angles.
Molecular geometry and shape of CIF3
We return to the Lewis structure of Chlorine Trifluoride to calculate its molecular geometry. Chlorine has an expanded octet, as shown from the Lewis structure. It is attached to two lone pairs.
Three Fluorine atoms also surround the central Chlorine atom. We can see the same thing and figure out the molecular geometry.
Fluorine atoms will resist each other, according to the VSEPR hypothesis. We get a Trigonal Planar geometry by putting the three Fluorine atoms on the centre Chlorine atom. The molecule geometry transforms to a T-shape when the two lone pairs of electrons come into the picture.
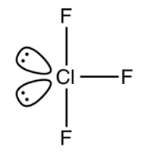
Trigonal Bipyramidal is the electronic geometry.
Conclusion
ClF3 hybridisation is sp3d. It has a T-shaped molecular geometry. There are 28 total valence electrons available to draw the ClF3 Lewis structure. It has a trigonal bipyramidal electron shape. According to the ClF3 Lewis structure, this molecule has two lone pairs and three bonded pairs.
 Profile
Profile Settings
Settings Refer your friends
Refer your friends Sign out
Sign out




