A chemist uses different types of notation or formulas to analyze a compound. These notations are empirical formulas, molecular formulas, and structural formulas. Different formulas are used for different purposes.
The empirical formula is the simplest one. It is the most straightforward one that is why it is also known as the analytics formula. They provide the lowest whole number ratio of the atoms of the elements present in an organic or inorganic compound. It presents the compositions of a compound. It does not show or provide the number of atoms of an element present in the compound; instead, it is the lowest possible whole number of the elements present in the compound.
The empirical formula is very useful for the primary purpose especially when all the data regarding the given compound is unavailable. It simplifies the complex sets of the various elements present in the compound. It gives information about the constituents of the compound at one glance.
Define Empirical Formula
The empirical formula of a compound can be defined as the lowest whole number ratio of all the atoms of the elements present in the compound. This is the formula that specifically represents the elements of an organic or inorganic compound. It does not provide more details about the compound like molecular formula. It is simply the formula of the elements which is written with the smallest subscript. The percent composition of a compound is related to the empirical formula of the compound.
The molecular formula provides information about the number of atoms of all the elements present in the compound. Whereas the structural formula is also such notation that shows the positioning of the atoms of different elements in the compound.
The empirical formula of a compound can be determined by using mass composition data. This process includes the following steps:
- An instrument called CHN is used to determine the composition of a compound. It can be used to calculate the mass fraction of different elements present in the given compound such as carbon, nitrogen, hydrogen, helium, oxygen etc.
- When the mass contribution elements are determined this information can be used to convert them into atoms.
- The empirical formula is the lowest possible whole number of the ratio between the atoms of elements in the compound.
Benzene Empirical Formula
Benzene is an organic compound that is made of carbon and hydrogen. There are six atoms of carbon and six atoms of hydrogen that together form benzene. The chemical formula of benzene is C6H6. It has only carbon and hydrogen as its constituents so it is also known as a hydrocarbon. Benzene is naturally found in crude oil which gives it a highly flammable property. It is among the top 20 production chemicals. It is used in the production of different products like rubber, nylon, lubricant, plastic, etc. However, it is hazardous to health. People who are exposed to it for a long time can get benzene-related diseases, even cancer too.
Some properties of benzene are as follows:
- It is a colorless or light yellow liquid.
- It is sweet in smell.
- It is made up of carbon and hydrogen.
- It is highly flammable.
As we discussed the molecular formula of benzene is C6H6 which indicates that it constitutes six carbon and six hydrogen atoms. When we find the ratio between the number of atoms of carbon and hydrogen we get 1:1. So the empirical formula of benzene is CH.
When there is no subscription or the numeric subscripts of any chemical symbol this means that there is the presence of 1 atom of that particular element. So, here we get 1-1 atom of each of the elements.
Empirical Formula against Molecular Formula
Empirical Formula is used to show what elements a compound is made of. By going through the empirical formula of a compound we can know what are the elements present in the given compound at a glance. The molecular formula of a compound will be important to know how many atoms of different elements are presented in a compound. Circumstances and needs will decide which formula is important.
Empirical Formula against Structural Formula
An empirical formula lacks some necessary details of the compounds. It does not provide any details regarding the structure of the atoms of different elements present in the compound. It just shows the representations of different elements with their simplest numeric values. Unlike empirical formula, the structural formula of a compound provides more details about the elements. It shows the positioning of the different atoms and their bonding with one another in the compound.
Empirical Formula Calculator:
Mass composition data can be a good way to determine the empirical formula of an element present in a compound. Using this method we can mathematically determine the ratio of the different elements present in a compound.
For this purpose, a CHN which is an instrument used by the chemist to determine the composition of an organic compound can be used to find the mass fraction of any element like hydrogen, carbon, oxygen, nitrogen, etc for any unknown organic compound.
Once the mass contribution of different elements is determined we can easily transform this information into the number of atoms present in the compound.
The empirical formula for the compound is determined by keeping the number of the atoms in their simplest or lowest form as the empirical formula is simply the lowest whole number ratio of the atoms of the elements present in the organic compound.
For the calculation of the empirical formula, we must, first of all, convert the mass percentage into grams. Thereafter the number of the moles should be calculated by using the given formula:
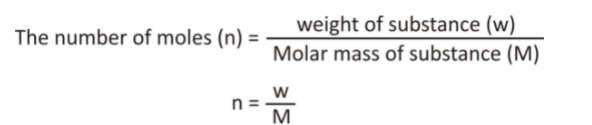
Then we have to calculate the simplest molar ratio of the subscriptions. For this we have to divide the moles we have already calculated by the smallest quotient. Now we have to calculate the lowest whole number ratio. The output is the empirical formula of the compound.
Conclusion
There are many notations that are used by a chemist to determine and describe atomic constituents in a symbolic way. These notations include empirical formulae, molecular formulae, and structural formulae. These formulae include the chemical symbols of different elements along with their numeric values which show their atomic composition in the given compound.
 Profile
Profile Settings
Settings Refer your friends
Refer your friends Sign out
Sign out





