We observe light around us in various colours. Did you know that the light we see is a form of energy? Also, what we see around us is not just limited to light. There are various other radiations we cannot notice. They all lie in the electromagnetic spectrum and are known as Electromagnetic Radiation.
Electromagnetic radiation is defined as the radiation that oscillates in space, in a direction mutually perpendicular to electrical and magnetic fields. The radiation can be of varying wavelengths, and they constitute the electromagnetic spectrum in the order of increasing wavelengths.
X-Rays
X-rays are high energy, high frequency, and low wavelength radiations. Because they have such high energy, they can penetrate through many objects, including skin and tissues. This makes them appropriate for taking a body scan, as they can penetrate through the skin, but get reflected by bones.
The wavelength of x-rays lies between 10-10 to 10-12 meters. The energy is high, ranging from 100eV to 100keV.
They were discovered in 1895 by Wilhelm Roentgen. At that time, their mystery could not be understood by scientists, hence the name ‘x-rays’. Now we know that, like all electromagnetic radiations, they have both particle and wave characters.
Atoms and X-rays notes
Now, we have a brief understanding of what X-rays are. Let’s move on to how they relate to atoms. Atoms consist of subatomic particles like electrons, photons, and neutrons. While photons and neutrons lie in the nucleus, electrons revolve around the nucleus in shells. The shells have varying energy levels.
So, whenever an electron makes a transition from n=1 (inner shell) to let’s say n=2 (outer shell), it requires energy to do so. Whenever it makes a transition from n=2 to n=1, it loses energy in the process. A photon is emitted, which has both particle and wave character. The wavelength of the photon is given by
𝝺 = hcE2 – E1
Where,
h is the Planck’s constant with a value of 6.6 x 10-34 Js
c= 3×108 m/s (speed of light)
E2 is the energy of electrons in the second shell
E1 is the energy of electrons in the first shell.
When an electron jumps from a much higher-level orbit to a lower-level orbit, the magnitude of the energy of the photon released is very high (because the energy difference between two such orbits is high). Correspondingly, radiation of a small wavelength, i.e., x-rays are emitted.
This simple understanding of atoms and x-rays helps us produce x-rays in an x-ray tube.
Production of X-rays
An x-ray tube comprises a glass chamber that has vacuum inside. Inside, there is a cathode (mainly of tungsten filament) that releases high-speed electrons. These high-speed electrons bombard the anode at the other end. The anode is made up of an element having a high atomic number, mostly Tungsten. Now, when high-speed electrons bombard the anode, they lose their energy in the form of heat and x-rays. 99% energy is lost to heat and the rest 1% to x-rays.
Two kinds of x-rays are produced- continuous and characteristic. The x-rays produced by the deceleration of incoming electrons when they strike the anode are called Continuous X-rays. This is because x-rays of several wavelengths are produced, as the energy lost by all striking electrons would not be the same.
Characteristic x-rays are produced when the incoming high-speed electron knocks off an anode electron from its shell, which leaves a vacancy in the shell from where the electron has been knocked off. That vacancy can be filled up by a higher-level anode electron, which jumps from a higher energy level to a lower energy level. In the process, it produces characteristic x-rays.
Atomic spectra
X-rays emitted from an atom can be labelled as K, L, M, N, and so on. These letters correspond to the energy level to which the electrons dropdown.
K corresponds to n=1 (first energy level). Whenever an electron from any of the higher energy levels falls to n=1, the K line is observed. If it is from n=2 to n=1, it would be K𝝰 x-rays. If it is from n=3 to n=1, it would be K𝝱 x-rays, and so on.
If the electron falls to n=2, it corresponds to the L line.
The wavelength of such x-rays for hydrogen and hydrogen-like systems can be calculated using the Rydberg formula:
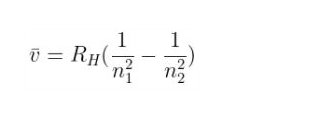
where,
RH is the Rydbegr’s constant with a value of 1.096 x 107 m-1
Z is the atomic number of the element
n12 is the square of the final energy level (its value is 1 in the case of K lines, 4 in the case of L lines, 9 for M lines, and so on).
n22 is the square of the initial energy level
It is clear that more is the atomic number, less is the wavelength or more is the frequency.
Conclusion
These atoms and X-rays JEE notes can help in the in-depth understanding of X-rays. They are a form of electromagnetic radiation which have high energy and can travel in vacuum. They are used to scan the body. Two kinds of x-rays, continuous and characteristic, are produced in the process. Characteristic X-rays have various spectral lines of differing energies and wavelengths, which are represented by alphabets K, L, M, N, and so on.
 Profile
Profile Settings
Settings Refer your friends
Refer your friends Sign out
Sign out





